by Joni Eronen

Nuclear magnetic resonance (NMR) is a powerful and versatile tool that is based on the observation of nuclear spin precession in a strong magnetic field. It is widely used in the field of environmental sciences research, such as in determining the three-dimensional structures of molecules as well as in medical imaging. Compared to other spectroscopic methods, NMR offers several benefits, such as exceptional chemical resolution, nondestructive and quantitative nature, and high reproducibility. However, it is notorious from its weak signal intensities.
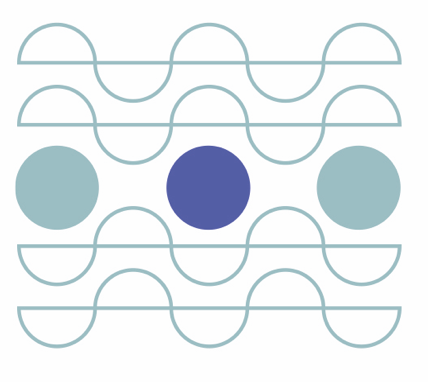
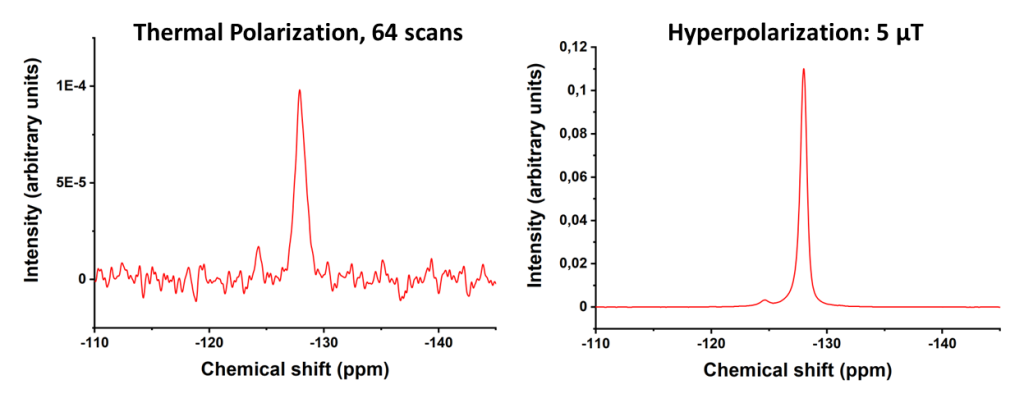
In NMR, the main reason behind its low signal intensity is the low spin polarization. When a sample is placed in a strong magnetic field, the individual spins can be thought of as small magnets that tend to align with the external magnetic field due to its lowest energy state, resulting in a macroscopic magnetization. However, in thermal equilibrium, the orientations of the spins follow the Boltzmann distribution, and, even at the highest magnetic fields available, the magnetic energy is tiny comparable to the thermal energy. This causes the orientations of the spins to be strongly disturbed by the thermal energy, resulting in a situation where most of the spins are oriented randomly. By making use of hyperpolarization methods, one can exceed the thermal polarization by several orders of magnitude, which significantly improves the NMR signal. Thermal polarization and hyperpolarization are demonstrated in Figure 1.
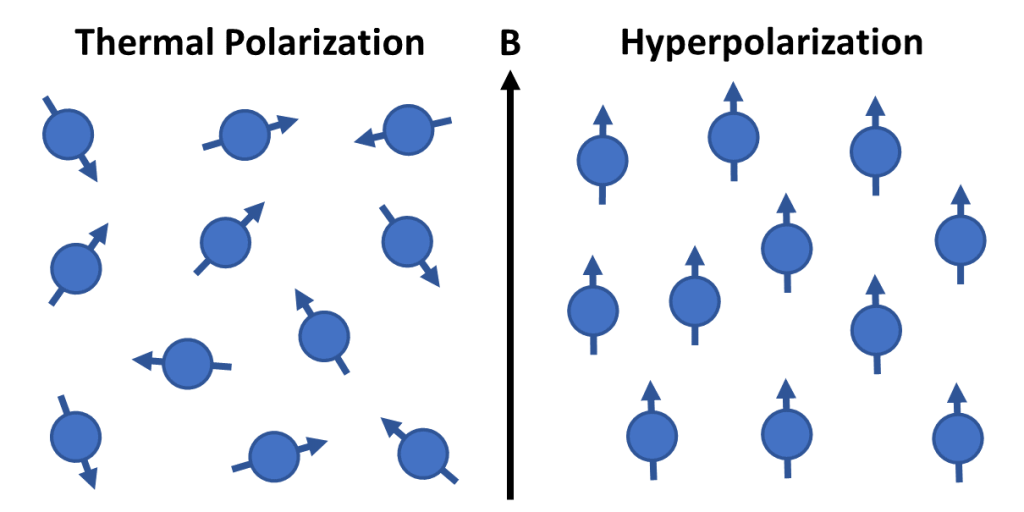
Signal amplification by reversible exchange (SABRE) is a recently discovered hyperpolarization method in which NMR signals can be enhanced by flowing parahydrogen through a sample in the presence of an organometallic catalyst. There are several advantages of using SABRE over other hyperpolarization methods. With SABRE, one can hyperpolarize any substrate that can transiently bind into the organometallic catalyst, there are no requirements for expensive equipment, making SABRE accessible to many users, and, in addition, the maximum obtainable polarization is achieved in the matter of seconds. Due to these promising features of SABRE, it has many future applications in the field of environmental sciences research as well as medical imaging.
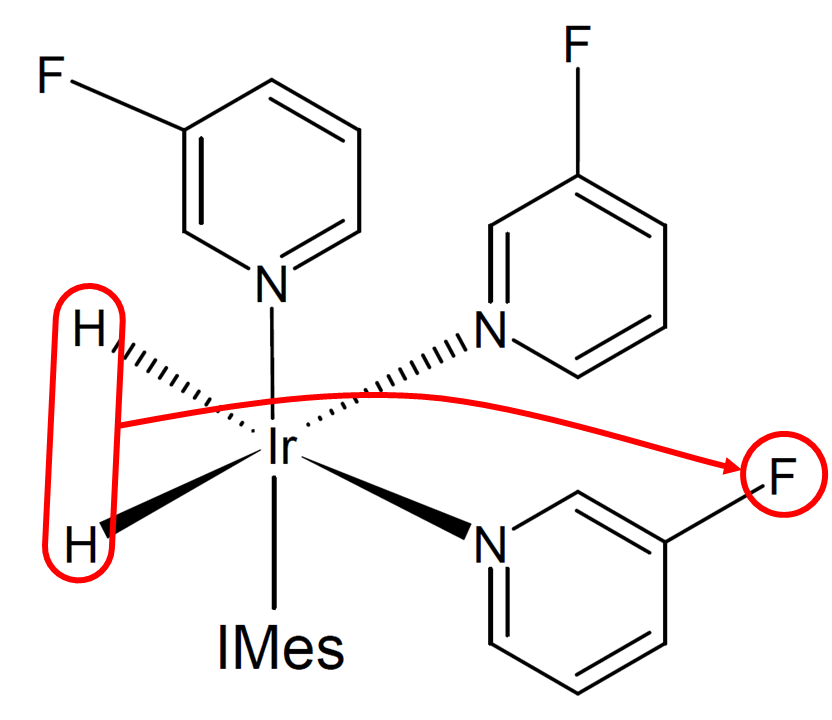
In SABRE, the source of hyperpolarization is the singlet state of dihydrogen, or in other words, parahydrogen. The spin order transfer from parahydrogen to the substrate occurs in an organometallic complex, shown in Figure 2, that is continuously formed and dissociated in the solution. There are different mechanisms that contribute to the spin order transfer in SABRE. Here, two main mechanisms, the coherent transfer through level anti-crossings (LACs) and the dipolar cross relaxation are briefly introduced. The minimal spin system that can be used to describe the coherent spin order transfer is formed by three spins, the dihydride protons and one spin that is in the bound substrate. If the polarizing magnetic field is tuned such that the resonance frequency difference between the dihydride protons and the substrate is equal to the scalar coupling between the dihydride protons, we are in a LAC condition, where the system starts to coherently oscillate between energy levels, as it is no longer in a true eigenstate. The LAC condition is in the millitesla range for 1H and in the microtesla range for other nuclei. If the system is in the LAC condition, and it is continuously exposed to fresh parahydrogen, the coherent oscillation will eventually cause the substrate to be hyperpolarized. The dipolar cross relaxation can be understood by considering a system of two spins. Because of dipolar coupling between the spins, the spins no longer relax individually. Instead, the rate of change of the spin 1 magnetization is affected by the magnetization of spin 2. This enables the spin order transfer from a spin to another.
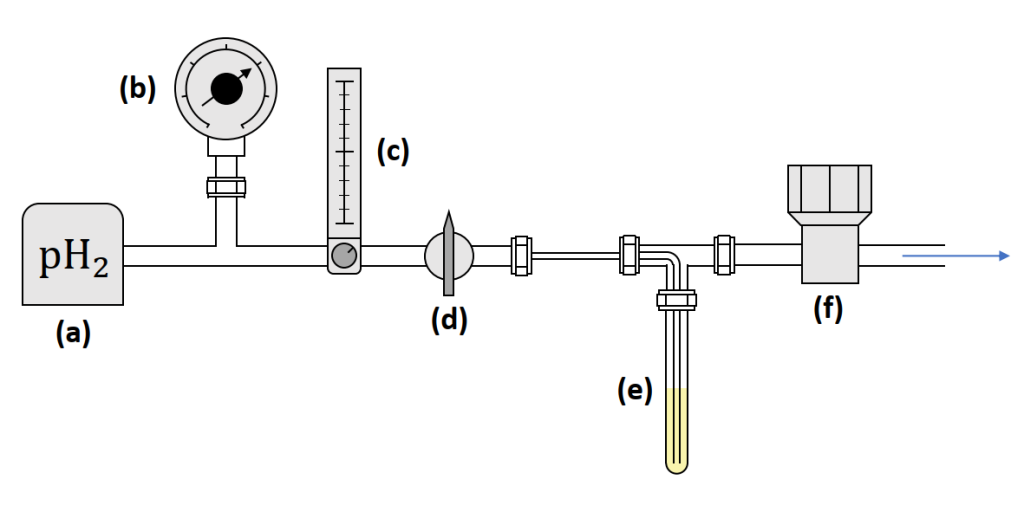
To obtain a better understanding of the different spin order transfer mechanisms in SABRE, in my master’s thesis work, several SABRE experiments with 3-fluoropyridine were carried out at the NMR laboratory in the University of Oulu. We studied the effect of polarizing field, and the relaxation and build-up times for both 19F and 1H nuclei. For this purpose, we built a hyperpolarization system in which parahydrogen could be flowed controllably through a liquid sample that is placed inside a regular 5 mm NMR tube. The system is shown schematically in Figure 3. To tune the polarization transfer field precisely in the microtesla range, we placed a self-made solenoid inside a Mu-Metal magnetic shield. In addition, we used a larger Helmholtz coil to produce fields in the millitesla range. The general procedure in the measurements was to insert the sample in the polarization transfer field, flow parahydrogen through the sample for a certain time, and transfer the sample to the spectrometer immediately after pausing the flow.
A significant increase in the polarization levels was observed for both nuclei. For 19F, the maximum enhancement factor over thermal polarization was 1406, corresponding to a polarization of 0.46%. As shown in Figure 4, this is a significant enhancement. The maximum signal was achieved when the polarizing field was set to 5 µT, which agrees with the value given by the LAC condition. In addition, there was a significant dependence on the polarizing field, indicating that the spin order transfer takes place through LACs. For 1H, the maximum enhancement was 2700, corresponding to a polarization of 0.94%. Again, a clear dependence of the polarizing field was observed with the maximum being at 8 mT, which also agrees with the LAC condition. However, since our Helmholtz coil was capped at 8 mT, concerning our data, we do not know whether greater levels of polarization could have been achieved using polarizing fields past 8 mT. Since significant enhancements were received at the LAC conditions for both nuclei, it was expected that a maximum for 19F would be found in the millitesla range due to dipolar cross relaxation from the 1H, and vice versa for the 1H in the microtesla range. However, there was no clear dependence to be seen based on the measurements, indicating that the cross relaxation between those nuclei would be ineffective.
Studying the relaxation and build-up times at different magnetic fields revealed that, for both nuclei, the relaxation is fast at ultralow fields (T1 ∼ 3 s), whereas it gets slower at higher fields (T1 ∼ 10 s). The build-up times turned out to be comparable to the relaxation times. The reason for faster relaxation at ultralow fields is that the molecular motions, which create fluctuating local magnetic fields, happen to match the resonance frequency. In addition, it is not surprising that the relaxation times are comparable between 19F and 1H, as dipolar coupling, which is likely the dominating relaxation mechanism at the magnetic fields under study, is comparable between the two nuclei due to their similar gyromagnetic ratios. In conclusion, this work demonstrated that SABRE can be used to effectively hyperpolarize 3-fluoropyridine. LACs were attributed as the main polarization transfer mechanism with the observation of maxima near the LAC conditions for both 19F and 1H, whereas relayed transfer through dipolar coupling was not observed for either nucleus. At the time of writing, using an improved hyperpolarization system, more systematic SABRE experiments with 3-fluoropyridine and 3,5-difluoropyridine are being performed to obtain further information about the spin order transfer process.